This Article
Citations

Except where otherwise noted, this work is licensed under Creative Commons Attribution-NonCommercial 4.0 International License.
Is Screening Imaging Necessary in Dilated Cardiomyopathy?
Abstract
Introduction: Dilated cardiomyopathy (DCM) is the leading cause of heart failure and arrhythmia.
Case Presentation: A 47-year-old male, diagnosed with dilated cardiomyopathy, died due to heart failure. During the screening of his family members, his 17-year-old daughter and 9-year-old son also had dilated cardiomyopathy. Another daughter had died suddenly at the age of 12 years.
Conclusions: We herein describe 3 patients with dilated cardiomyopathy developing in the father, daughter, and son of the same family and justify the importance of the screening test as an important tool for identifying families affected by familial dilated cardiomyopathy.
Keywords: Dilated Cardiomyopathy; Familial; Genetic; Cardiac Imaging; Echocardiography
1. Introduction
Dilated cardiomyopathy (DCM) is the leading cause of heart failure and arrhythmia. There are many possible causes for the dilatation and dysfunction of the heart such as coronary artery disease, infection, and excessive use of alcohol. In cases where the cause of DCM is unknown, the condition is called idiopathic DCM. A family history of DCM is found in about one-third to one-half of patients with idiopathic DCM (1), who are considered to have familial DCM. Familial DCM is commonly reattached to the genetic mutations that affect myocardial functions. We report three cases of DCM developing in the father, daughter, and son of the same family.
2. Case Presentation
A 47-year-old man was brought to our emergency room for shortness of breath, dizziness, and palpitation. He had been previously diagnosed with non-ischemic DCM but was on irregular medications. On physical examination, the patient was ill and restless with dyspnea (New York Heart Association functional class IV). His blood pressure could not be recorded, and his heart rate was 130 beats/minute. Intravenous inotropic agents were started. However, he developed bradycardia and asystole and expired despite receiving CPR.
One month later, his 17-year-old daughter was referred for cardiac evaluation. She presented with difficulty in breathing of 5 months' duration, associated with bilateral pitting pedal edema. She denied a previous history of any kind of illness, fever, and joint pain; and she was not under any medications. On examination, she had dyspnea, with a blood pressure of 100/60 mmHg and a pulse rate of 105 beats/minute. Complete blood count and serum biochemistry were within normal limits. Electrocardiography showed sinus tachycardia, and chest X-ray revealed cardiomegaly. Thereafter, transthoracic echocardiography demonstrated global left ventricular (LV) hypokinesia, grossly dilated LV [LV ejection fraction (LVEF) of 31%] and left atrium, and severe mitral regurgitation (Figure 1).
|
Figure 1.
Echocardiography (Parasternal Long-Axis View) of a 17-Year-Old Girl, Showing a Grossly Dilated Left Ventricle (LVIDd = 76.6 mm and LVIDs = 64.7 mm) and Left Atrium with a Left Ventricular Ejection Fraction of 31% and Fractional Shortening of 15.5%
|
We advised the other family members to undergo echocardiographic screening to rule out DCM. During evaluation, the girl's younger brother, a previously healthy 9-year-old child with no cardiovascular history, had a history of dyspnea on moderate exertion. Cardiovascular examination revealed apex beat at the sixth intercostal space at the level of the anterior axillary line. There was a grade II pan-systolic murmur at the apex, radiating to the axilla. The LV third heart sound was audible. Chest X-ray showed cardiomegaly. The boy, subsequently, underwent transthoracic echocardiography, which revealed global LV hypokinesia, grossly dilated LV (LVEF = 20%) and left atrium, and severe mitral regurgitation (Figure 2).
|
Figure 2.
Echocardiography (Parasternal Long-Axis View) in a 9-Year-Old Boy, Showing a Grossly Dilated Left Ventricle (LVIDd = 65.0 mm and LVIDs = 59.2 mm) and Left Atrium with a Left Ventricular Ejection Fraction of 20% and Fractional Shortening of 8.9%
|
Careful history taking revealed that another younger daughter of the family had died suddenly 2 years previously at the age of 12 years. The mother and the elder son were screened by electrocardiography as well as echocardiography and found to be normal. We diagnosed a case of familial DCM in the father, elder daughter, and younger son as well as the possibility of DCM in the younger daughter, who had died suddenly at 12 years of age. The affected children were managed with guideline-directed medical therapy for heart failure with a reduced EF such as diuretics (including Spironolactone), angiotensin-converting enzyme (ACE) inhibitors, and cardioselective beta blockers. The unaffected elder son was advised to undergo serial screening on a 3 to 5-year basis.
3. Discussion
DCM differs from other forms of the secondary dilatation and dysfunction of the ventricles due to known cardiac or systemic processes. These are referred to as specific cardiomyopathies, named after the disease process with which they are associated, such as ischemic cardiomyopathy, valvular heart cardiomyopathy, hypertensive cardiomyopathy, alcoholic cardiomyopathy, and myocarditis. Less common forms of specific cardiomyopathies are peripartum cardiomyopathy; cardiomyopathies developing in patients with amyloidosis, hemochromatosis, and sarcoidosis; and cardiomyopathies due to toxicity from agents like Doxorubicin.
If no specific cause for DCM is known, it is termed "idiopathic DCM" and the disease is defined as familial if there are at least two idiopathic DCM diagnoses in the closely related family members or a relative of a DCM patient with unexplained sudden death before the age of 35 years (2, 3). Familial DCM is caused by mutations in the genes that encode cytoskeletal and sarcomeric proteins in cardiac myocytes. At least 50 single genes have been identified to be linked to familial DCM. The majority of these elicit disease as dominant mutations (4); however, they collectively may account for only a fraction of genetic causation. It is anticipated that additional genes causative of familial DCM will be discovered.
Also, an autosomal dominant pattern is the main mode of transmission for DCM, accounting for 70–90% of familial cases (1). In a minority of cases (5–10%), DCM may be due to a gene mutation on the X chromosome. These X-linked conditions, including Becker and Duchene muscular dystrophies, usually affect only males; daughters of affected males will be carriers of the condition as they will have inherited the faulty gene on the X chromosome from their father and they may be mildly affected (1). More rarely, the condition may be inherited as an autosomal recessive disorder (5), where a mutation is inherited from both parents, or as a mutation in the mitochondrial DNA which is passed on in the ovum.
The screening test for the first-degree relatives of idiopathic DCM patients has been recommended for identifying families affected by familial DCM. Proper history taking and echocardiographic screening of the first-degree relatives of individuals with familial DCM are indicated, which may permit early diagnosis and treatment before the onset of advanced symptomatic disease (6). Serial screening may also be indicated on a 3 to 5 year basis for otherwise normal but at-risk family members (6). More frequent screening can be advised to those with mild abnormalities or unexplained symptoms. In families in which familial DCM has an onset only in adults, it may be reasonable to wait until the early teenage years for a full evaluation, including echocardiography (6).
According to the recent guidelines, genetic testing may be considered in conjunction with genetic counseling (7). However, genetic testing for familial DCM is not routinely available. Genetic testing is also complicated by the locus and allelic heterogeneity as well as the very low prevalence rates of most familial DCM genes identified to date, making genetic testing insensitive (1, 5)Finally, patients with familial DCM are treated with the same guideline-directed medical therapy such as diuretics (including Spironolactone), ACE inhibitors, and cardioselective beta blockers, which are used to treat other forms of DCM and heart failure. Patients who present with conduction-system disturbances should be followed up for arrhythmias that might necessitate permanent pacemaker implantation in later life (8). Individuals with syncopal episodes and/or a strong family history of sudden death may require electrophysiological studies and automated implantable cardioverter-defibrillator implantation (8).
Currently, no trials exist on the usefulness of therapy in asymptomatic affected patients. However, it is believed that in patients who are affected but asymptomatic, an early use of ACE inhibitors and/or beta blockers could be very important in slowing the progression of the disease (6). Patients with severe heart failure, severe reduction in the functional capacity, and depressed LVEF, despite guideline-directed medical therapy, have a low survival rate and may require heart transplantation (6). Screening imaging is recommended every 3 to 5 years in adults or children who are the first-degree relatives of an individual with idiopathic DCM.
References
- 1. Taylor MR, Carniel E, Mestroni L. Cardiomyopathy, familial dilated. Orphanet J Rare Dis. 2006;1:27. [DOI] [PubMed]
- 2. Mestroni L, Rocco C, Gregori D, Sinagra G, Di Lenarda A, Miocic S, et al. Familial dilated cardiomyopathy: evidence for genetic and phenotypic heterogeneity. Heart Muscle Disease Study Group. J Am Coll Cardiol. 1999;34(1):181-90. [PubMed]
- 3. McNally EM, Golbus JR, Puckelwartz MJ. Genetic mutations and mechanisms in dilated cardiomyopathy. J Clin Invest. 2013;123(1):19-26. [DOI] [PubMed]
- 4. Burkett EL, Hershberger RE. Clinical and genetic issues in familial dilated cardiomyopathy. J Am Coll Cardiol. 2005;45(7):969-81. [DOI] [PubMed]
- 5. Schonberger J, Seidman CE. Many roads lead to a broken heart: the genetics of dilated cardiomyopathy. Am J Hum Genet. 2001;69(2):249-60. [DOI] [PubMed]
- 6. Hershberger RE, Morales A, Siegfried JD. Clinical and genetic issues in dilated cardiomyopathy: a review for genetics professionals. Genet Med. 2010;12(11):655-67. [DOI] [PubMed]
- 7. Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Jr, Drazner MH, et al. 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;62(16):e147-239. [DOI] [PubMed]
- 8. Fatkin D, members of the CCGDCWG. Guidelines for the diagnosis and management of familial dilated cardiomyopathy. Heart Lung Circ. 2011;20(11):691-3. [DOI] [PubMed]
 Home
Home Archive
Archive Search
Search Sign In
Sign In Site Menu
Site Menu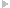 Email this article to a friend
Email this article to a friend





